Introducing Chemical Bonding
Chemical bonds form when the electrons of two or more atoms interact. The atoms will either share or transfer electrons to make each of the interacting atoms more stable, creating ionic or covalent bonds. 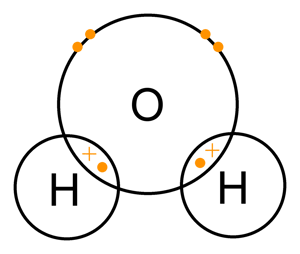
When simplifying this concept to explain to students where chemical energy is stored, you can use the analogy of two magnets interacting rather than presenting the complex concepts of bonding. Atoms creating a chemical bond can be thought of as two magnets attracting. The positive pole of one magnet is attracted to the negative pole of the other magnet, just like a positively charged atom is attracted to a negatively charged atom.
When two oppositely charged atoms move close together, like the magnets, they may naturally stick together to become more stable. After the magnets are joined, energy (a force applied over a distance) is necessary to separate the magnets.
In a similar way, energy is required to break a chemical bond that joins two atoms together in the bond of a molecule. The creation of a chemical bond releases chemical energy as atoms become more stable. Breaking chemical bonds requires chemical energy as atoms require energy to be forced from a stable configuration. A chemical reaction involves breaking bonds of the reactants and creating bonds of the new products. Whether a chemical reaction yields a net absorption or release of energy depends upon the specific reaction.
Featured Kit: Elementary Energy
